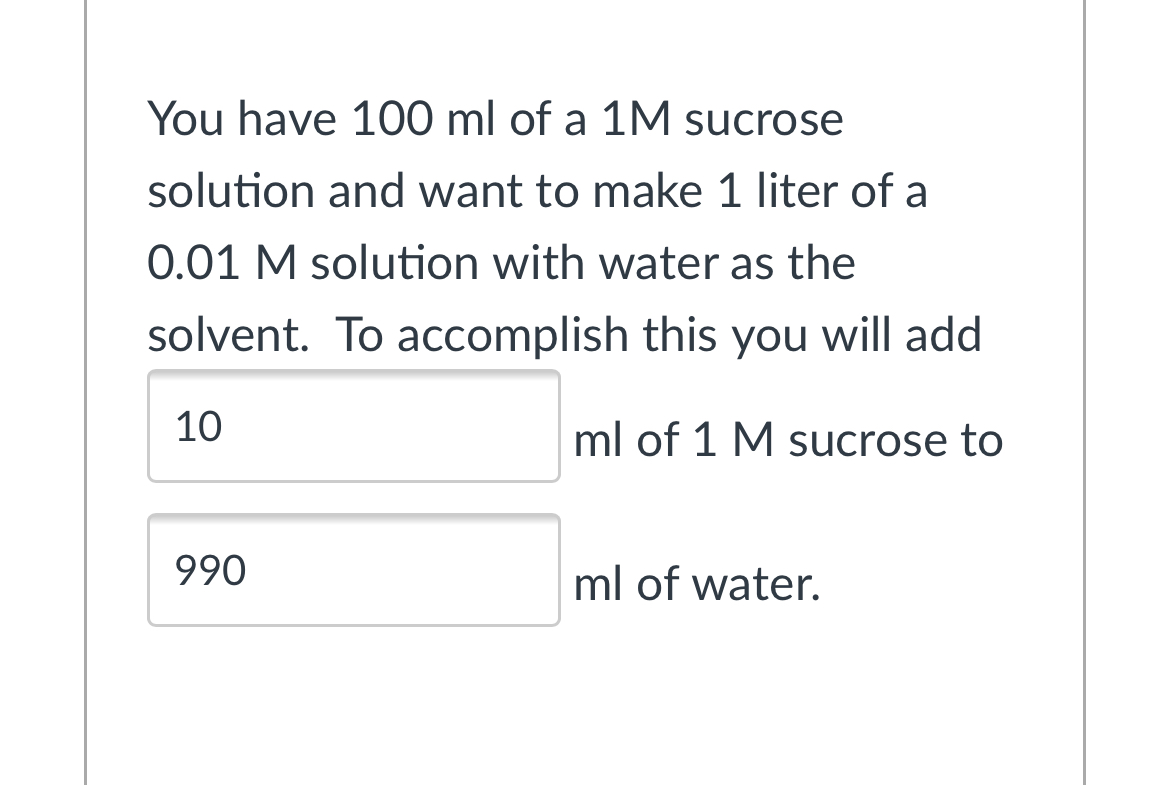
Calculate the degree of hydrolysis of the 0.01 M solution of salt` (KF)(Ka(HF)=6.6xx10^(-4))` :- - YouTube
What is the pH of the resulting solution when the equal volume of 0.1M Noah and 0.01M HCL are mixed? - Quora

pH Meter, Dr.meter Upgraded 0.01 Resolution High Accuracy PH Tester with Backlit Two-Color LCD Display and ATC, 0-14pH Measurement Range with Data Hold Function Digital pH Tester Pen-PH838: Amazon.com: Industrial & Scientific

It has been found that the `pH` of a `0.01 M` solution of an organic acid is `4.15`. Calculate t... - YouTube

Welcome to Chem Zipper.com......: Aqueous solution of 0.004 M Na2SO4 and 0.01 M glucose are iosotonic .The degree of dissociation of Na2SO4 is ..
The specific conductance of 0.01M solution of KCl is 0.0014per ohm cm at 25^°C .It's equivalent conductance is